Molecule Splitting Process and Technology Explained
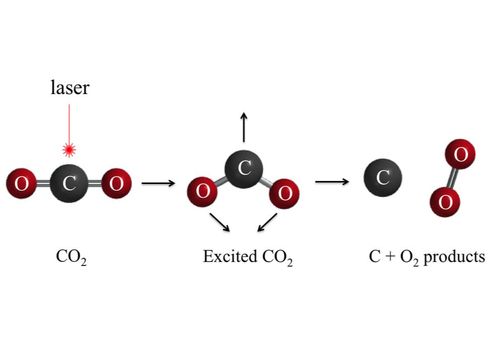
In our CCC device, there is a chamber in which CO2 molecules get separated into Carbon and 2 parts Oxygen. In chemistry, normally molecules split in a way they lose the least energy, meaning CO2 will break into one oxygen atom and a carbon monoxide molecule (CO). Since Carbon is double bonded to the Oxygen atoms, it would require much less energy to break one oxygen atom bond instead of two. Also, CO (Carbon Monoxide) is more stable than regular Carbon, making that another reason CO2 will split up this way. In order to avoid this, by giving the CO2 molecule more energy, the molecule will start to “roam”, which is a chemical phenomenon where chemical bonds break up in different ways. By doing this, CO2 will be separated into Carbon and 2 parts Oxygen, which is what we want. When CO2 molecules enter this chamber, they are fired with a HEVUVL (High Energy Vacuum Ultraviolet Laser), splitting them into Carbon and 2 parts Oxygen. The Oxygen is released back into the environment, while the carbon is used for carbon products plastics and graphene (The thinnest, strongest material known).